Asked by Maria Castro on Jul 31, 2024

Verified
When propylene reacts with hydrogen bromide in the presence of a peroxide initiator, which of the following structures are formed during the mechanism?
A) 
B) 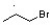
C) 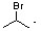
D) H∙
Peroxide Initiator
A compound that decomposes to form free radicals, initiating polymerization reactions.
Propylene
A flammable gas that is a colorless hydrocarbon used as a fuel and a petrochemical feedstock.
- Identify reactants, products, and intermediates in reaction mechanisms involving alkenes.

Verified Answer
FH
Fatin Husna Amat SahriAug 06, 2024
Final Answer :
B
Explanation :
The mechanism for the reaction of propylene with hydrogen bromide in the presence of a peroxide initiator involves the formation of a free radical intermediate. The correct structure formed during the mechanism is 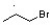 , which shows the free radical intermediate where the bromine has added to the carbon with the most hydrogen atoms. The other structures listed are not involved in the mechanism. H∙ is a hydrogen radical and is not a product of this reaction.
, which shows the free radical intermediate where the bromine has added to the carbon with the most hydrogen atoms. The other structures listed are not involved in the mechanism. H∙ is a hydrogen radical and is not a product of this reaction.
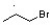 , which shows the free radical intermediate where the bromine has added to the carbon with the most hydrogen atoms. The other structures listed are not involved in the mechanism. H∙ is a hydrogen radical and is not a product of this reaction.
, which shows the free radical intermediate where the bromine has added to the carbon with the most hydrogen atoms. The other structures listed are not involved in the mechanism. H∙ is a hydrogen radical and is not a product of this reaction.
Learning Objectives
- Identify reactants, products, and intermediates in reaction mechanisms involving alkenes.
Related questions
Draw a Chair Conformation of the Starting Substituted Cyclohexane Molecule ...
How Many Distinct Alkene Products Results When 1-Bromo-1-Ethylcyclopentane Is Treated ...
Which of the Following Intermediates Is Thought to Occur in ...
Dehydrohalogenation of an Alkyl Halide by Treating It with a ...
Provide the Reagents Necessary for Carrying Out the Transformation of ...