Asked by Hebah Shihadeh on Sep 27, 2024

Verified
How many nodes, other than the node coincident with the molecular plane, are found in the highest energy π MO of 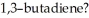
A) 0
B) 1
C) 2
D) 3
E) none of the above
Energy π MO
Refers to molecular orbitals formed by the overlap of pi orbitals, affecting molecular stability and reactivity, particularly in conjugated systems.
- Gain an understanding of the theory behind molecular orbitals and how they influence dienes' stability.

Verified Answer
BC
Berto Castellanosabout 2 hours ago
Final Answer :
D
Explanation :
The highest energy π MO of the given molecule has two nodes not coincident with the molecular plane. One node occurs along the x-axis and another occurs along the y-axis. Therefore, there are 3 nodes in total (including the node coincident with the molecular plane).

Learning Objectives
- Gain an understanding of the theory behind molecular orbitals and how they influence dienes' stability.