Asked by Gurwinder Bhullar on Jun 04, 2024

Verified
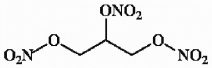
Nitroglycerine is formed by the reaction of glycerine (propane-1,2,3-triol) and nitric acid. Provide the structure of nitroglycerine.
Nitroglycerine
A highly volatile and explosive chemical compound used in dynamite and in medicinal preparations as a vasodilator.
Glycerine
A simple polyol compound used in foods, pharmaceuticals, and cosmetics for its moisturizing and solvent properties.
Nitric Acid
A highly corrosive and strong mineral acid with the formula HNO3, commonly used in the production of fertilizers, explosives, and in various nitration processes.
- Comprehend the fundamental principles of organic reactions and their processes.
- Distinguish among energy materials by examining their chemical configurations and responses.

Verified Answer

Learning Objectives
- Comprehend the fundamental principles of organic reactions and their processes.
- Distinguish among energy materials by examining their chemical configurations and responses.
Related questions
Provide the Name of the Major Organic Product That Results ...
The Williamson Ether Synthesis Proceeds Via an ________ Mechanism ...
What Class of Organic Compound Is the Major Product of ...
Given a Sample of (R)-2,3-Dimethylhexan-3-Ol, Which of the Following Would ...
Provide a Mechanism for the Reaction Shown Below