Asked by Santiago Talamantes on May 28, 2024

Verified
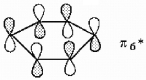
Show how the participating p orbitals interact to form the highest energy π molecular orbital of benzene.
P Orbitals
Atomic orbitals shaped like dumbbells, consisting of regions where there's a high probability of finding an electron outside the nucleus, pivotal in chemical bonding and valence.
Highest Energy
Refers to the state of a system or particle where it possesses the maximum amount of energy compared to other possible states.
- Understand the relationship between molecular orbital theory and the chemical behavior of aromatic compounds.

Verified Answer
AM

Learning Objectives
- Understand the relationship between molecular orbital theory and the chemical behavior of aromatic compounds.
Related questions
In the Molecular Orbital Representation of Benzene, How Many π ...
How Many Distinct Nodal Planes Which Are Perpendicular to the ...
What Is the Bond Order of the Carbon-Carbon Bonds in ...
An Ylide Is a Molecule That Can Be Described as ...
From a Molecular Orbital Perspective Why Isn't There Relatively Free ...