Asked by Lazaro Arozarena on Sep 29, 2024
The protons marked Ha and Hb in the molecule below are ________. 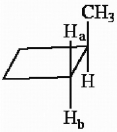
A) chemically equivalent
B) enantiotopic
C) diastereotopic
D) endotopic
E) none of the above
Chemically Equivalent
Term used to describe atoms or groups in a molecule that have identical chemical environments and exhibit the same chemical behavior.
Diastereotopic
Describes hydrogen atoms or groups attached to the same carbon atom that are not equivalent in the three-dimensional spatial configuration, leading to non-superimposable structures.
Enantiotopic
Referring to atoms or groups in a molecule that are mirror images of each other but are not superimposable, often relating to prochiral centers.
- Identify the various proton settings in a molecule.

Learning Objectives
- Identify the various proton settings in a molecule.
Related questions
Use the Structure Below to State the Relationship Between the ...
The Protons Marked H a and H b in the Molecule Below ...
The Compound Methylamine, CH 3 NH 2 , Contains a C-N Bond ...
Which of the Following Molecules Contains a Polar Covalent Bond ...
What Are the Relative Integrations for the 1 H NMR Signals ...