Asked by Brittany Lavender on Jun 19, 2024

Verified
The synthetic steroid RU-486 is shown below. How many pi bonds does RU-486 contain? 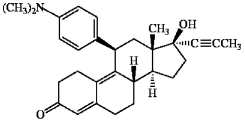
Pi Bonds
Covalent chemical bonds where two lobes of one involved atomic orbital overlap two lobes of the other involved atomic orbital, forming a bond that is parallel to the bonding axis and allowing for electron sharing.
RU-486
A synthetic steroid used medically to induce abortion by blocking the action of progesterone.
- Determining the number of sigma and pi bonds present in molecules.

Verified Answer
JL

Learning Objectives
- Determining the number of sigma and pi bonds present in molecules.
Related questions
What Two Atomic Orbitals or Hybrid Atomic Orbitals Overlap to ...
UV Spectroscopy Measures the Energy Required to Promote an Electron ...
In the UV-Visible Spectrum of (E)-1,3,5-Hexatriene, the Lowest Energy Absorption ...
A Molecule of Sodium Chloride (NaCl) Is ____ Because Each ...
Which Type of Bond Is Formed Whenever Atoms Share One ...