Asked by Trista Adams on May 28, 2024

Verified
Which H atom in the molecule shown will be most readily abstracted by a bromine radical? 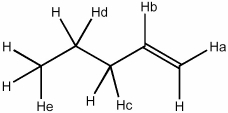
A) Ha
B) Hb
C) Hc
D) Hd
E) He
Bromine Radical
A highly reactive species formed by the homolytic cleavage of bromine, involved in various combustion and halogenation processes.
Abstracted
In chemistry, this term refers to the concept of removing something, especially a significant component or element from a complex entity.
- Explain the basic principles of free radical mechanisms, encompassing the initiation, propagation, and termination stages.

Verified Answer
SN
Sairyl NavascaJun 01, 2024
Final Answer :
C
Explanation :
The H atom on the carboxyl group (Hc) will be most readily abstracted by a bromine radical because it is the most weakly bonded to the molecule due to the electron-withdrawing effect of the nearby carbonyl group. This weakens the bond between the H atom and the neighboring O atom, making it more susceptible to abstraction by a radical.

Learning Objectives
- Explain the basic principles of free radical mechanisms, encompassing the initiation, propagation, and termination stages.
Related questions
Which of the Halogens Below Undergoes Free Radical Halogenation with ...
How Many Distinct Monochlorinated Products Can Result When Cyclopentane Is ...
What Is the Name of the Major Monobrominated Product Which ...
Which of the Following Is Not a Possible Termination Step ...
When the Reaction Between Methane and Chlorine Is Photochemically Initiated ...