Asked by Shedly Fevrier on Sep 28, 2024

Verified
Which of the following structures (a-d) is another resonance structure of the following organic molecule? 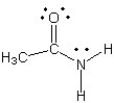
A) 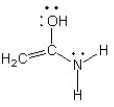
B) 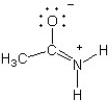
C) 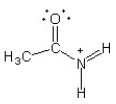
D) 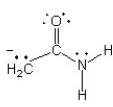
Resonance Structure
Different Lewis structures representing the same molecule that differ only in the positions of the electrons, not the positions of the atoms.
Organic Molecule
A molecule of the kind typically found in living systems, often containing carbon atoms bonded to hydrogen, oxygen, or nitrogen.
- Draft precise Lewis structures for molecules and ions, with resonance forms included as applicable.
- Perceive and clarify the essentiality of resonance in the stability of molecular structures.

Verified Answer
CS
Carlos Santana-sigarretaabout 17 hours ago
Final Answer :
B
Explanation :
The only difference between the given structure and option B is the location of a double bond. This relocation of the double bond results in a change of electron distribution and delocalization, making it a resonance structure. Option A and D have different overall structures and option C does not have any changes in double bond positions.

Learning Objectives
- Draft precise Lewis structures for molecules and ions, with resonance forms included as applicable.
- Perceive and clarify the essentiality of resonance in the stability of molecular structures.
Related questions
When a Negatively Charged Species Is Most Appropriately Depicted as ...
Structures ________, Shown Below, Are Resonance Structures, and Structure ________ ...
Nitroamines Are Common Functional Groups Found in Energetic Materials, Such ...
When a Molecule Can Best Be Represented as a Series ...
Provide the Major Resonance Structures of the Ion Which Results ...