SD
shubham deshmukh
Answers (7)
SD
Answered
HBr can be added to an alkene in the presence of peroxides (ROOR) . What function does the peroxide serve in this reaction?
A) nucleophile
B) electrophile
C) radical chain initiator
D) acid catalyst
E) solvent
A) nucleophile
B) electrophile
C) radical chain initiator
D) acid catalyst
E) solvent
On Aug 01, 2024
C
SD
Answered
Of the following, who is the only modern scientist?
A) Socrates
B) Thomaford
C) Democritus
D) Dalton
A) Socrates
B) Thomaford
C) Democritus
D) Dalton
On Jul 10, 2024
D
SD
Answered
Explain why SN2 reactions on allyl bromide proceed faster than corresponding reactions on ethyl bromide.
On Jun 10, 2024
When the substrate is allylic, resonance stabilization through conjugation with the adjacent π bond occurs in the transition state. This delocalization lowers the energy of the transition state and results in an enhanced rate.
SD
Answered
Deduce the identity of the following compound from the 1H NMR data given.
C3H6Br2: δ 2.4 (2H, quintet), 3.5 (4H, triplet) (ppm)
C3H6Br2: δ 2.4 (2H, quintet), 3.5 (4H, triplet) (ppm)
On Jun 07, 2024
BrCH2CH2CH2Br
SD
Answered
In the lock and key model of molecular structure, what term is used for the "key" molecule?
A) The antigen
B) The antibody
C) The antibacterial
D) The antibiotic
A) The antigen
B) The antibody
C) The antibacterial
D) The antibiotic
On Jun 03, 2024
A
SD
Answered
Describe the major products of the following reaction and predict which of the products would predominate. Explain and defend your answer. 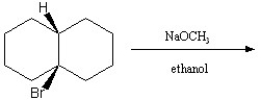

On May 31, 2024
Possible products: 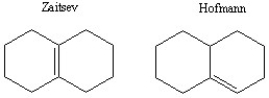 The Hofmann product would most likely predominate due to the coplanar cis configuration of the Br and H across the bridge. The E2 mechanism favors anti alignment of the H with the halogen leaving group. There are two possible anti-aligned H atoms that would lead to the Hofmann product.
The Hofmann product would most likely predominate due to the coplanar cis configuration of the Br and H across the bridge. The E2 mechanism favors anti alignment of the H with the halogen leaving group. There are two possible anti-aligned H atoms that would lead to the Hofmann product.
 The Hofmann product would most likely predominate due to the coplanar cis configuration of the Br and H across the bridge. The E2 mechanism favors anti alignment of the H with the halogen leaving group. There are two possible anti-aligned H atoms that would lead to the Hofmann product.
The Hofmann product would most likely predominate due to the coplanar cis configuration of the Br and H across the bridge. The E2 mechanism favors anti alignment of the H with the halogen leaving group. There are two possible anti-aligned H atoms that would lead to the Hofmann product.SD
Answered
Provide the proper IUPAC name for the alkene shown below. 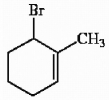

On May 28, 2024
6-bromo-1-methylcyclohexene